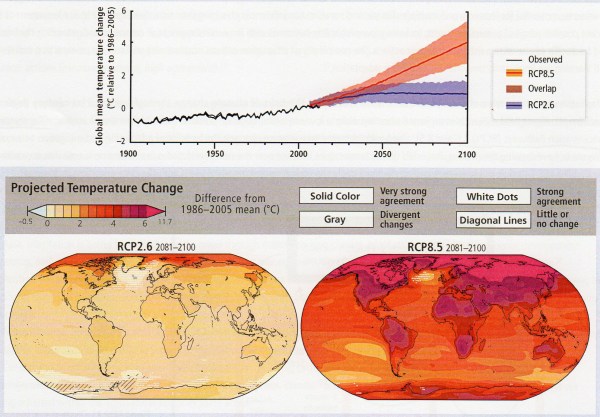
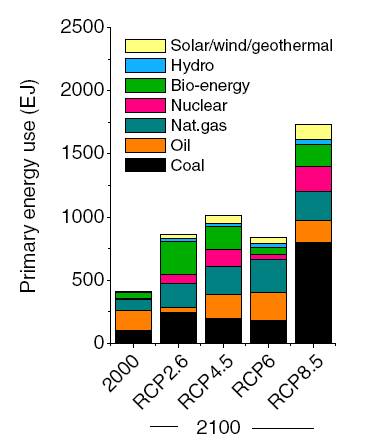
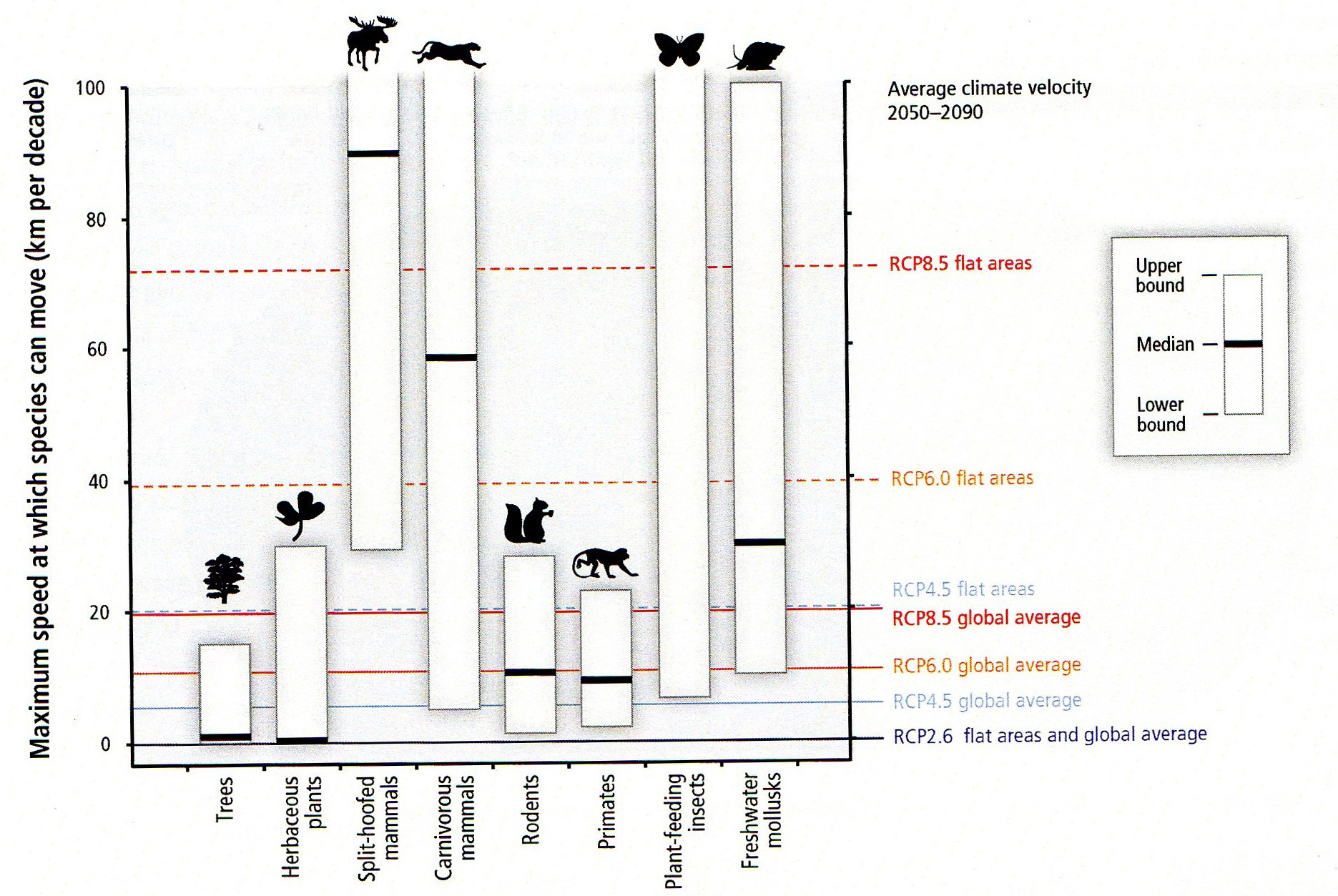
I was encouraged to hear a presentation by an academic scientist at the recent Beyond Pesticides Forum that was another indication of the paradigm shift in invasive species management toward a less destructive approach. Dr. Bernd Blossey is a Professor at Cornell University, where he directs the Ecology and Management of Invasive Plants Program in the Department of Natural Resources. His many years of studying invasive plants, such as purple loosestrife, garlic mustard, water chestnut, Japanese knotweed, and phragmites have convinced him that there are often “multiple stressors” that contribute to such invasions. Some factors such as the presence of earthworms and deer can be more important factors in the Northeast than the non-native plants themselves.
Based on his research experience, Dr. Blossey delivered wise advice to land managers at the Beyond Pesticide Forum. The featured photo at the top of this article was his introductory slide.
Before a restoration project begins, these questions should be asked and answered:

If the project seems worthwhile after such analysis is done, this is Dr. Blossey’s advice about monitoring the project and measuring its success:

Practicing what he preaches
Dr. Blossey used these principles in his study of garlic mustard in the forests of the northeast. (1) Over a period of more than 10 years, Dr. Blossey and his collaborators measured the abundance of garlic mustard in 16 plots from New Jersey to Illinois where no attempt had been made to control or eradicate it. They found that growth rates initially increased, but decreased over time and eventually the population started to decline. Dr. Blossey explained their findings in a recent webinar that is available HERE:

Garlic mustard was first recorded in North America in 1868 on Long Island, New York. It spread west from there and is now found from southern Canada to Georgia and from New York and Quebec to Oregon, British Columbia and Alaska. Because land managers believed that garlic mustard suppresses populations of native plants, they have been trying to eradicate garlic mustard in northern forests for decades, with little long term success. Dr. Blossey addressed that concern in his webinar.

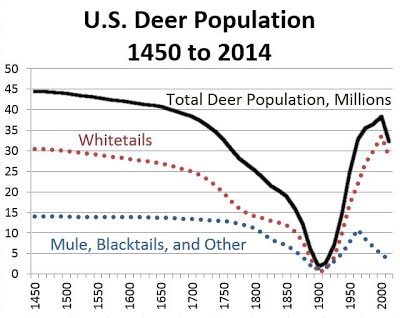
Earthworms are the prerequisite for garlic mustard invasion. Earthworms in northern forests are also considered alien invaders because they were killed, along with forests, by advancing glaciers during the Ice Age. When forests returned after the Ice Age over 10,000 years ago, they evolved without earthworms that were reintroduced by European settlers less than 500 years ago.
When deer are excluded from areas by fencing plots with garlic mustard populations, abundance of native vegetation does not decline. Deer have a strong preference for native vegetation. Absent deer, garlic mustard does not seem to suppress the growth of native plants in northern forests.
In other words, garlic mustard is not guilty as charged. Dr. Blossey explains the disadvantages of attempting to eradicate it. The decline of garlic mustard abundance over time is attributed to negative soil feedback that builds over time as the soil microbial community responds to the new plant. Removing garlic mustard episodically prolongs the process of building that negative soil feedback. When groups of well-meaning young people are sent into the forest to pull garlic mustard, they trample the very native plants they are trying to save.
Are there lessons for land managers in the Bay Area?
Because garlic mustard doesn’t exist in California and our native earthworms are considered beneficial to soil health, you might wonder if this study is relevant here. California was not glaciated during the Ice Age. Our earthworms survived the Ice Age and they evolved with our forests.
So, what can we learn from this study? The pattern of initial growth and eventual decline of populations of introduced plants is not unique to garlic mustard: “A phenomenon that has received increased attention is whether introduced species go through boom and bust cycles, ultimately becoming non-threatening members of local communities.” (1) One recently published study was based on nearly 5,000 vegetation inventories collected in 49 National Parks in the eastern United States. It reported that non-native plants appeared to decline after 100-200 years:
“Residence time appears a core part of invasion that interacts with other mechanisms, such as climate matching, propagule pressure and empty niche. Initially, time appears to benefit non-native species as they establish in a novel range. They likely face low enemy loads, and any successful dispersal increases their populations and invaded range. As they spread, initial barriers, such as distance or suboptimal habitat, were overcome, as was resistance from native relatives. However, their biggest challenge appeared to be time, as they all declined after ~1 to 2 centuries, suggesting that pathogens and herbivores caught up with them.” (2)
The message for land managers everywhere is that patience is needed to judge the impact of introduced species. Most will fit into ecosystems eventually and attempts to speed up that process often do more harm than good. We can’t judge changes in nature by the short-term perspective of human lifetimes because the evolution of nature is a continual process that began long before humans existed and is likely to persist long after we are gone.
Applying Dr. Blossey’s “Core Knowledge” to local projects
What if Dr. Blossey’s “Core Knowledge” had been applied to projects in the San Francisco Bay Area? Here are examples of local eradication projects that might have benefitted:
- San Francisco has been trying to eradicate oxalis in its parks for over 20 years by spraying a selective herbicide (Garlon). There seems to be more oxalis now than there was 20 years ago. Oxalis is visible only about 2 months of the year. When it dies back in the spring it leaves behind the native plants with which it co-exists. If a control plot had been set aside before they started eradicating oxalis perhaps we would know the answer to these important questions: Does oxalis suppress the growth of native plants? Does attempting to eradicate oxalis produce more or less oxalis?
- California, Oregon, and Washington have been trying to eradicate non-native spartina marsh grass along the entire West Coast for over 20 years. Here in the Bay Area, non-native species of spartina have been 99% eradicated, but a hybrid of the native and the non-native remains and is poisoned with imazapyr annually. According to a recent presentation by the Invasive Spartina Project, the hybrid is visually indistinguishable from the native and it occupies the same elevation of the marsh. Over 500 genetic tests are needed every year to distinguish the hybrid from the native in order to poison the hybrid. Dr. Blossey’s approach might ask these important questions: What harm is hybrid spartina doing? Do more or fewer animals live in hybrid spartina? What effect has 20 years of spraying imazapyr had on the soil and the microbes that live in it? Is the eradication project doing more harm than good?

We don’t know the answers to these important questions because projects were initiated and implemented without the analysis and monitoring metrics needed to answer the questions. The projects continue without being accountable for the damage they are doing. Public money is funding these projects without requiring the projects to be accountable for the consequences.
California has made a commitment to spend billions of dollars on “nature based solutions” and achieving “biodiversity goals.” This is an opportunity to start new projects off on the right foot by:
- Requiring the analysis needed to determine the impacts and causes of perceived problems in the environment.
- Requiring control plots so that the effects of the project can be compared with the option of not doing the project.
- Requiring that projects be monitored, using established metrics so that the success of the project can be measured.